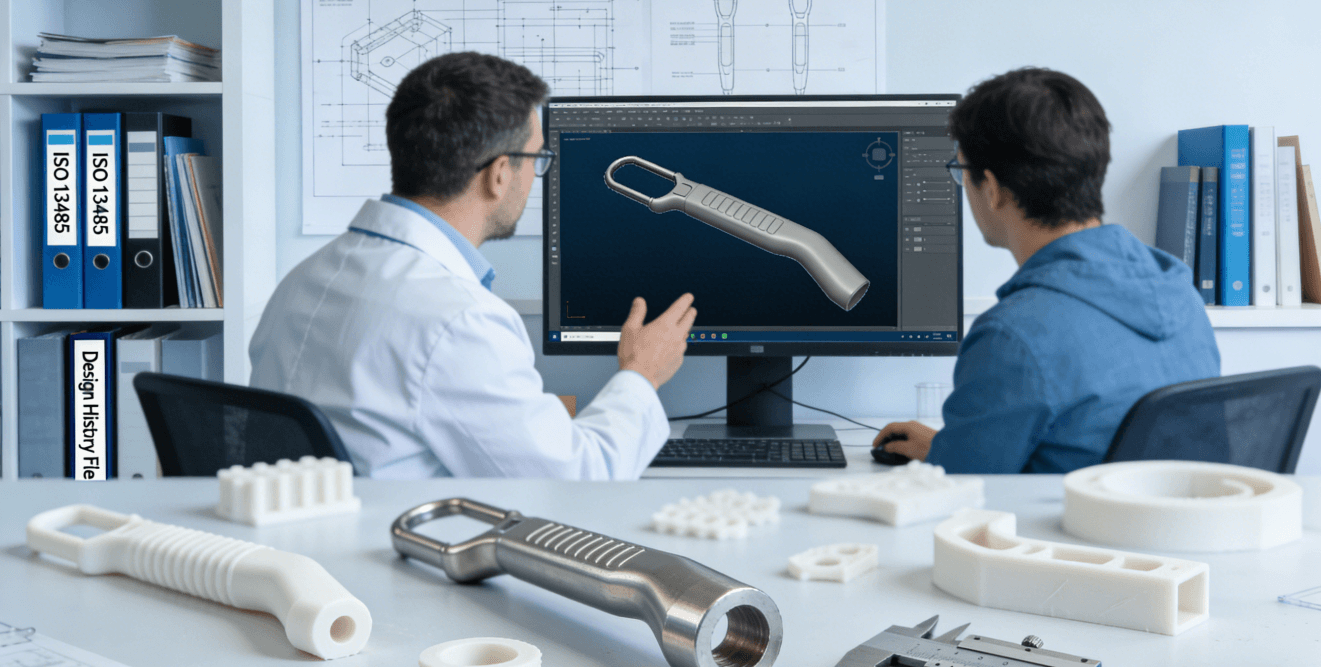
Medical Device Prototyping Behind the 80% Failure Rate—How to Avoid Compliance Pitfalls with the FDA and ISO 13485
Introduction For medtech startups and R&D teams, translating an innovative concept into a physical prototype ready for testing and regulatory scrutiny is a critical, high-risk phase. Many teams invest months of effort and significant resources, only to face major setbacks—or even failure—on the eve of animal trials or regulatory submission due to issues with material …